
|
시장보고서
상품코드
2003241
가금류 진단 시장 : 제품 유형별, 검사 유형별, 질병 유형별, 가금 유형별, 검체 유형별 - 세계 예측(-2031년)Poultry Diagnostics Market by Product Type (Consumable & Reagent, Instrument), Test Type (PCR, ELISA, Rapid Antigen Tests), Disease Type, Poultry Type (Broiler, Layer, Turkey, Duck & Geese), Sample Type (Swabs, Serum, Blood) - Global Forecast to 2031 |
||||||
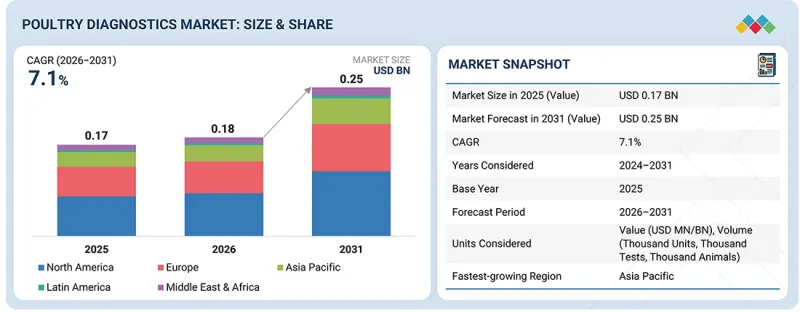
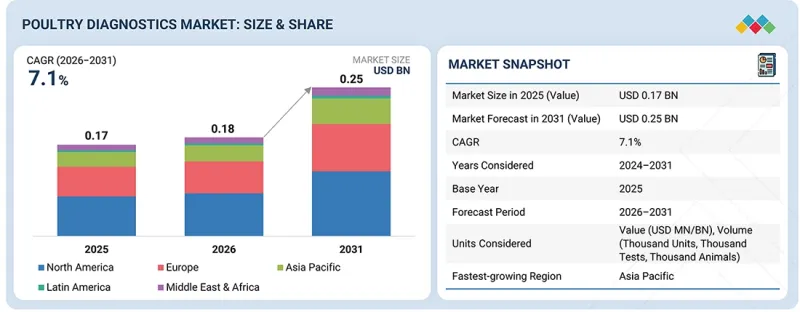
가금류 진단 시장 규모는 2026년 1억 8,000만 달러에서 2031년까지 2억 5,000만 달러로 성장할 것으로 예상되며, CAGR은 7.1%가 되면 예측됩니다.
이러한 성장은 가금류 질병 진단, 모니터링 및 양계장 건강 관리의 미래를 형성하는 몇 가지 주요 요인에 의해 주도되고 있습니다. 가금류 진단 시장의 주요 촉진요인은 전 세계 상업용 가금류 생산의 급속한 확대와 대규모 가금류 사육에서 전염병 발생을 억제하고자 하는 수요 증가입니다.
| 조사 범위 | |
|---|---|
| 조사 대상 기간 | 2024-2031년 |
| 기준 연도 | 2025년 |
| 예측 기간 | 2025-2031년 |
| 대상 단위 | 금액(달러) |
| 부문 | 유형, 제형, 적응증, 모달리티, 투여 경로, 용도, 최종사용자, 지역 |
| 대상 지역 | 북미, 유럽, 아시아태평양, 라틴아메리카, 중동, 아프리카 |
가금류 사육이 더욱 집약화됨에 따라, 축산 사업자들은 생산 손실을 방지하고 양계의 지속적인 생산성을 보장하기 위해 질병의 조기 발견과 정기적인 건강 모니터링을 필수적인 전략으로 도입하고 있습니다. 그 결과, 가금류 농장, 수의학 실험실, 통합형 가금류 기업은 조류인플루엔자, 뉴캐슬병, 전염성 기관지염 등의 병원체를 신속하고 정확하게 식별하기 위해 PCR, ELISA와 같은 고급 진단법을 점점 더 많이 활용하고 있습니다.
수요를 이끄는 또 다른 중요한 요인은 전 세계 가금류 공급망 전반에 걸쳐 바이오 안전, 식량 안보, 규제 대상 질병 관리에 대한 관심이 높아지고 있다는 점입니다. 각국 정부, 국제기구, 양계업자들은 육류와 계란 생산에 막대한 손실을 초래하고 국제 무역을 혼란에 빠뜨릴 수 있는 대규모 질병 발생에 대비하기 위해 질병 추적 시스템을 강화하고 있습니다. 그 결과, 생산 시설에 대한 안전한 접근을 보장하고, 생산자와 수의학 당국이 안정적인 생산 수준을 유지하면서 적시에 봉쇄 조치를 취할 수 있도록 가금류 검사에 진단 기술을 활용하는 것이 일반적인 관행이 되었습니다.
또한, 분자진단, 자동화 및 대량 검사 능력의 발전으로 가금류 질병 식별정확성과 효율성이 크게 향상되었습니다. 신속 PCR 검사, 로봇을 이용한 ELISA 조작, 휴대용 진단 키트, 첨단 검체 처리 등의 혁신 기술을 통해 대응 시간을 단축하고 전체 양떼에 대한 대규모 검사가 가능해졌습니다. 이러한 기술적 진보를 통해 가금류 생산자와 진단 검사 기관은 질병 관리 전략을 강화하고, 양계장의 건강 상태를 최적으로 모니터링하며, 현대 가금류 사육 시스템에서 생태계의 안정성을 촉진할 수 있습니다.
"검사 유형별로는 PCR 기반 진단 부문이 예측 기간 동안 가장 높은 CAGR로 성장할 것으로 예상됩니다."
PCR 기반 진단 부문은 높은 민감도와 특이성, 병원균을 신속하게 검출할 수 있는 능력으로 인해 가금류 진단 시장에서 가장 높은 성장세를 보일 것으로 예상됩니다. PCR 기술은 바이러스나 박테리아의 유전물질이 미량이라도 식별이 가능합니다. 이 기능을 통해 조류인플루엔자, 뉴캐슬병, 전염성 기관지염과 같은 가금류 질병을 증상 발현 전 단계에서 감지할 수 있어 적시에 개입할 수 있는 기회를 제공할 수 있습니다. 조기 개입은 전염병의 확산과 영향을 억제하고 생산 손실을 줄이는 데 도움이 됩니다. 또한, 집약적 가금류 생산 시스템에서 신속하고 대규모 검사에 대한 수요 증가와 질병 모니터링의 향상으로 인해 상업용 농장 및 수의학 진단 연구소에서 PCR 기반 진단의 보급이 촉진되고 있습니다.
"검체 유형별로는 면봉이 2025년 가장 큰 시장 점유율을 차지했습니다."
2025년, 면봉은 채취의 용이성, 비침습성, 가금류 병원체 검출에 대한 높은 신뢰성으로 인해 가금류 진단 시장을 주도했습니다. 조류독감, 뉴캐슬병, 전염성 기관지염 등의 질병에 대해 채취하는 가장 일반적인 두 종류의 면봉 검체는 구강인두 면봉과 배설구 면봉입니다. 이 면봉 검체는 질병 감시 및 발병 조사에 자주 사용됩니다. 면봉은 PCR 및 ELISA와 같은 널리 사용되는 진단 방법과 호환되기 때문에 가금류 농장, 수의학 실험실 및 모니터링 프로그램에서 선호되는 검체 유형입니다. 또한, 면봉은 많은 수의 닭에서 시료를 쉽게 채취할 수 있어 대규모 검사 및 모니터링 프로그램에 적합하며, 이는 가금류 질병 진단에 광범위하게 사용되는 이유를 설명해줍니다.
"아시아태평양은 예측 기간 동안 가장 높은 성장률을 보일 것으로 예상됩니다."
아시아태평양은 가금류 진단 시장에서 가장 빠르게 성장하고 있는 지역입니다. 이러한 성장은 가금류 생산량 급증, 소비율 증가, 중국, 인도, 동남아시아 국가 등 주요 가금류 생산국에서의 질병 감시 확대 등 여러 요인에 기인합니다. 이 지역에서는 조류인플루엔자와 뉴캐슬병이 자주 발생하여 조기 발견과 정기적인 가금류 모니터링의 중요성이 부각되고 있습니다. 또한, 수의학 인프라의 개선과 질병 관리 및 식품 안전에 대한 정부 주도의 노력이 확대됨에 따라, 이 지역의 가금류 농장 및 수의학 연구소는 첨단 진단 기술을 보다 신속하게 도입할 수 있게 되었습니다.
세계의 가금류 진단 시장을 조사했으며, 시장 개요, 시장 성장에 영향을 미치는 각종 영향요인 분석, 기술·특허 동향, 법·규제 환경, 사례 분석, 시장 규모 추정 및 예측, 각종 부문별·지역별·주요 국가별 상세 분석, 경쟁 구도, 주요 기업 개요 등의 정보를 정리하여 전해드립니다.
자주 묻는 질문
목차
제1장 소개
제2장 주요 요약
제3장 주요 인사이트
제4장 시장 개요
제5장 업계 동향
제6장 기술, 특허, 디지털 및 AI의 도입에 의한 전략적 디스럽션
제7장 지속가능성과 규제 상황
제8장 고객 상황과 구매 행동
제9장 가금류 진단 시장 : 제품 유형별
제10장 가금류 진단 시장 : 검사 유형별
제11장 가금류 진단 시장 : 질병 유형별
제12장 가금류 진단 시장 : 가금 유형별
제13장 가금류 진단 시장 : 검체 유형별
제14장 가금류 진단 시장 : 최종사용자별
제15장 가금류 진단 시장 : 지역별
제16장 경쟁 구도
제17장 기업 개요
제18장 조사 방법
제19장 부록
KSM 26.04.27The poultry diagnostics market is projected to grow from USD 0.18 billion in 2026 to USD 0.25 billion by 2031, at a CAGR of 7.1%. This growth is driven by several key factors that are shaping the future of poultry disease diagnostics, surveillance, and flock health management. The primary driver of the poultry diagnostics market is the rapid expansion of commercial poultry production worldwide and the increasing demand to control infectious disease outbreaks in large flocks.
| Scope of the Report | |
|---|---|
| Years Considered for the Study | 2024-2031 |
| Base Year | 2025 |
| Forecast Period | 2025-2031 |
| Units Considered | Value (USD billion) |
| Segments | Type, Form, Indication, Modality, Route of Administration, Application, End User, and Region |
| Regions covered | North America, Europe, Asia Pacific, Latin America, and Middle East, and Africa |
As poultry farming becomes more intensive, businesses in animal husbandry are adopting early disease detection and regular health monitoring as essential strategies to prevent production losses and ensure the ongoing productivity of their flocks. Consequently, poultry farms, veterinary laboratories, and integrated poultry companies are increasingly using advanced diagnostic methods, such as PCR and ELISA, to quickly and accurately identify pathogens, including avian influenza, Newcastle disease, and infectious bronchitis.
Another significant factor driving demand is the heightened focus on biosafety, food security, and the control of regulated diseases throughout the global poultry supply chain. Governments, international organizations, and poultry farmers are enhancing their disease-tracking systems to prepare for major outbreaks that could result in substantial losses in meat and egg production and disrupt international trade. As a result, the use of diagnostics in poultry testing has become a common practice to facilitate safe access to production facilities and enable producers and veterinary authorities to implement timely containment measures while maintaining stable production levels.
Moreover, advancements in molecular diagnostics, automation, and mass testing capabilities have significantly improved the accuracy and efficiency of identifying poultry diseases. Innovations such as rapid PCR testing methods, robotic ELISA operations, portable diagnostic kits, and advanced sample processing are reducing response times and enabling large-scale testing of entire flocks. By leveraging these technological advancements, poultry producers and diagnostic laboratories can enhance their disease control strategies, ensuring optimal flock health monitoring and promoting ecological stability within modern poultry farming systems.
"By test type, the PCR-based diagnostics segment is projected to grow at the highest CAGR during the forecast period."
The PCR-based diagnostics segment is expected to experience the highest growth in the poultry diagnostics market due to its high sensitivity and specificity, as well as its ability to quickly detect pathogens. PCR technology can identify even trace amounts of viral or bacterial genetic material. This capability allows for the detection of poultry diseases, such as avian influenza, Newcastle disease, and infectious bronchitis, at the pre-symptomatic stage, providing a timely opportunity for intervention. Intervening early helps limit the spread and impact of epidemics, reducing production losses. Additionally, the increasing demand for rapid, large-scale testing and improved disease monitoring in intensive poultry production systems is driving the widespread adoption of PCR-based diagnostics on commercial farms and in veterinary diagnostic laboratories.
"By sample type, the swabs segment accounted for the largest market share in 2025."
In 2025, swabs led the poultry diagnostics market due to their ease of collection, non-invasive nature, and high reliability in detecting poultry pathogens. The two most common types of swab samples collected for diseases such as avian influenza, Newcastle disease, and infectious bronchitis are oropharyngeal and cloacal swabs. These swab samples are often used for disease surveillance and outbreak investigations. Swabs are the preferred sample type on poultry farms, in veterinary laboratories, and in surveillance programs because they are compatible with widely used diagnostic methods such as PCR and ELISA. Additionally, swabs facilitate sample collection from large flocks, making them ideal for large-scale testing and monitoring programs, which explains their extensive use in poultry disease diagnostics.
"The Asia Pacific region is expected to witness the highest growth rate during the forecast period."
The Asia Pacific region is experiencing the fastest growth in the poultry diagnostics market. This growth can be attributed to several factors, including the rapid increase in poultry production, higher consumption rates, and the expansion of disease surveillance in major poultry-producing countries such as China, India, and various Southeast Asian nations. The region has faced multiple outbreaks of avian influenza and Newcastle disease, highlighting the importance of early detection and regular flock monitoring. Additionally, improvements in veterinary infrastructure and the rise of government initiatives for disease control and food safety are enabling poultry farms and veterinary laboratories in the region to adopt advanced diagnostic technologies more swiftly.
Breakdown of supply-side primary interviews:
By Company Type: Tier 1 (60%), Tier 2 (30%), and Tier 3 (10%)
By Designation: C-level Executives (30%), Directors (50%), and Other Designations (20%)
By Region: North America (40%), Europe (25%), Asia Pacific (20%), Latin America (10%), and the Middle East & Africa (5%)
Breakdown of demand-side primary interviews:
By End User: Veterinary Diagnostic Laboratories (45%), Integrated Poultry Producers (24%), Government & Reference Laboratories (14%), Academic & Research Institutions (10%), and Other Specialized Laboratories (7%)
By Designation: Heads/Directors of Veterinary Diagnostic Laboratories (47%), Poultry Veterinarians/Technical Services Managers (22%), Animal Health Surveillance Officers (15%), and Others (16%)
By Region: North America (25%), Europe (24%), Asia Pacific (25%), Latin America (11%), and the Middle East & Africa (15%)
Research Coverage
The market study covers the poultry diagnostics market in various segments. It aims to estimate the market size and growth potential by product type, test type, disease type, poultry type, sample type, end user, and region. The study also includes an in-depth competitive analysis of the market's key players, along with their company profiles, key observations on their products and business offerings, recent developments, and key market strategies.
Reasons to Buy the Report
The report can assist established companies and newer or smaller firms in understanding market trends, enabling them to capture a larger market share. Firms that acquire the report can implement one or more of the five strategies outlined below.
This report provides insights into the following points:
- Analysis of key drivers (rising incidence of transboundary & endemic diseases and growth in commercial & industrial poultry production), restraints (high cost of advanced molecular testing platforms and limited diagnostic infrastructure in developing regions), opportunities (geographic expansion in emerging poultry markets and development of rapid point-of-care (POC) tests), and challenges (rapid mutation and strain variability of viral pathogens and limited awareness among small & backyard farmers) influencing the growth of the poultry diagnostics market.
- Product Development/Innovation: Detailed insights on upcoming technologies and product launches in the poultry diagnostics market.
- Market Development: Comprehensive information about lucrative emerging markets. The report analyzes the markets for various types of poultry diagnostic products across regions.
- Market Diversification: Exhaustive information about products, untapped regions, recent developments, and investments in the poultry diagnostics market.
- Competitive Assessment: In-depth assessment of market shares, strategies, products, distribution networks, and manufacturing capabilities of the leading players in the poultry diagnostics market.
TABLE OF CONTENTS
1 INTRODUCTION
- 1.1 STUDY OBJECTIVES
- 1.2 MARKET DEFINITION
- 1.3 STUDY SCOPE
- 1.3.1 MARKET SEGMENTATION & REGIONAL SCOPE
- 1.3.2 INCLUSIONS & EXCLUSIONS
- 1.3.3 YEARS CONSIDERED
- 1.4 MARKET STAKEHOLDERS
- 1.5 SUMMARY OF CHANGES
2 EXECUTIVE SUMMARY
- 2.1 MARKET HIGHLIGHTS & KEY INSIGHTS
- 2.2 KEY MARKET PARTICIPANTS: MAPPING OF STRATEGIC DEVELOPMENTS
- 2.3 DISRUPTIVE TRENDS IN POULTRY DIAGNOSTICS MARKET
- 2.4 HIGH-GROWTH SEGMENTS
- 2.5 REGIONAL SNAPSHOT: MARKET SIZE, GROWTH RATE, AND FORECAST
3 PREMIUM INSIGHTS
- 3.1 POULTRY DIAGNOSTICS MARKET OVERVIEW
- 3.2 ASIA PACIFIC: POULTRY DIAGNOSTICS MARKET, BY TEST TYPE & COUNTRY (2025)
- 3.3 POULTRY DIAGNOSTICS MARKET: REGIONAL MIX
- 3.4 POULTRY DIAGNOSTICS MARKET: GEOGRAPHIC GROWTH OPPORTUNITIES
- 3.5 POULTRY DIAGNOSTICS MARKET: DEVELOPED VS. EMERGING ECONOMIES
4 MARKET OVERVIEW
- 4.1 INTRODUCTION
- 4.2 MARKET DYNAMICS
- 4.2.1 DRIVERS
- 4.2.1.1 Rising incidence of transboundary and endemic diseases
- 4.2.1.2 Growth in commercial and industrial poultry production
- 4.2.1.3 Shift toward preventive flock health management
- 4.2.1.4 Technological advancements in molecular diagnostics, multiplex assays, and rapid testing platforms
- 4.2.2 RESTRAINTS
- 4.2.2.1 High cost of advanced molecular diagnostic platforms and automated systems
- 4.2.2.2 Limited diagnostic infrastructure in developing regions
- 4.2.3 OPPORTUNITIES
- 4.2.3.1 Geographic expansion in emerging poultry markets
- 4.2.3.2 Expansion of rapid point-of-care and portable on-farm diagnostic solutions
- 4.2.4 CHALLENGES
- 4.2.4.1 Rapid mutation and strain variability of viral pathogens
- 4.2.4.2 Limited awareness among small & backyard farmers
- 4.2.1 DRIVERS
- 4.3 UNMET NEEDS & WHITE SPACES
- 4.3.1 UNMET NEEDS IN POULTRY DIAGNOSTICS MARKET
- 4.3.2 WHITE SPACE OPPORTUNITIES
- 4.4 INTERCONNECTED MARKETS & CROSS-SECTOR OPPORTUNITIES
- 4.4.1 INTERCONNECTED MARKETS
- 4.4.2 CROSS-SECTOR OPPORTUNITIES
- 4.5 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
- 4.5.1 STRATEGIC MOVES BY TIER-1/2/3 PLAYERS
5 INDUSTRY TRENDS
- 5.1 PORTER'S FIVE FORCES ANALYSIS
- 5.1.1 THREAT OF NEW ENTRANTS
- 5.1.2 THREAT OF SUBSTITUTES
- 5.1.3 BARGAINING POWER OF SUPPLIERS
- 5.1.4 BARGAINING POWER OF BUYERS
- 5.1.5 INTENSITY OF COMPETITIVE RIVALRY
- 5.2 MACROECONOMIC INDICATORS
- 5.2.1 INTRODUCTION
- 5.2.2 GDP TRENDS & FORECAST
- 5.2.3 TRENDS IN GLOBAL HEALTHCARE INDUSTRY
- 5.2.4 TRENDS IN GLOBAL ANIMAL HEALTH INDUSTRY
- 5.3 SUPPLY CHAIN ANALYSIS
- 5.4 VALUE CHAIN ANALYSIS
- 5.5 ECOSYSTEM ANALYSIS
- 5.6 PRICING ANALYSIS
- 5.6.1 AVERAGE SELLING PRICE TREND OF POULTRY DIAGNOSTIC PRODUCTS, BY KEY PLAYER, 2023-2025
- 5.6.2 AVERAGE SELLING PRICE TREND OF POULTRY DIAGNOSTIC PRODUCTS, BY REGION, 2023-2025
- 5.6.2.1 Average selling price trend of ELISA kits & reagents, by region, 2023-2025
- 5.6.2.2 Average selling price trend of PCR reagents & kits, by region, 2023-2025
- 5.6.2.3 Average selling price trend of sample preparation & extraction kits, by region, 2023-2025
- 5.6.2.4 Average selling price trend of controls & laboratory consumables, by region, 2023-2025
- 5.6.2.5 Average selling price trend of PCR instruments, by region, 2023-2025
- 5.6.2.6 Average selling price trend of ELISA readers & washers, by region, 2023-2025
- 5.6.2.7 Average selling price trend of nucleic acid extraction systems, by region, 2023-2025
- 5.7 TRADE ANALYSIS
- 5.7.1 TRADE DATA FOR HS CODE 3822
- 5.7.1.1 Import data for HS Code 3822
- 5.7.1.2 Export data for HS Code 3822
- 5.7.1 TRADE DATA FOR HS CODE 3822
- 5.8 KEY CONFERENCES & EVENTS, 2026-2027
- 5.9 TRENDS/DISRUPTIONS IMPACTING CUSTOMERS' BUSINESSES
- 5.10 INVESTMENT & FUNDING SCENARIO
- 5.11 SUCCESS STORIES & REAL-WORLD APPLICATIONS
- 5.11.1 AI-POWERED POULTRY DISEASE DIAGNOSTICS FOR COCCIDIOSIS & SALMONELLA DETECTION
- 5.11.2 MOLECULAR DETECTION AND CHARACTERIZATION OF AVIAN PATHOGENS USING MULTIPLEX PCR
- 5.11.3 MULTIPLEX PCR DETECTION OF RESPIRATORY PATHOGENS IN COMMERCIAL POULTRY FLOCKS
- 5.12 IMPACT OF 2025 US TARIFFS ON POULTRY DIAGNOSTICS MARKET
- 5.12.1 INTRODUCTION
- 5.12.2 KEY TARIFF RATES
- 5.12.3 PRICE IMPACT ANALYSIS
- 5.12.4 IMPACT ON COUNTRY/REGION
- 5.12.5 IMPACT ON END-USE INDUSTRIES
- 5.12.5.1 Veterinary diagnostic laboratories
- 5.12.5.2 Integrated poultry producers
- 5.12.5.3 Government & reference laboratories
- 5.12.5.4 Academic & research institutions
- 5.12.5.5 Other specialized laboratories
6 STRATEGIC DISRUPTIONS THROUGH TECHNOLOGY, PATENTS, AND DIGITAL & AI ADOPTION
- 6.1 TECHNOLOGY ANALYSIS
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.1.1.1 CRISPR-based molecular diagnostics
- 6.1.1.2 Isothermal amplification and paper-based LAMP
- 6.1.2 COMPLEMENTARY TECHNOLOGIES
- 6.1.2.1 Environmental & wastewater surveillance
- 6.1.2.2 Microfluidic sensors
- 6.1.3 ADJACENT TECHNOLOGIES
- 6.1.3.1 AI & machine learning
- 6.1.3.2 Multiplex PCR panels
- 6.1.1 KEY EMERGING TECHNOLOGIES
- 6.2 TECHNOLOGY/PRODUCT ROADMAP
- 6.2.1 NEAR TERM (2025-2027)
- 6.2.2 MID TERM (2028-2030)
- 6.2.3 LONG TERM (2030+)
- 6.3 PATENT ANALYSIS
- 6.3.1 PATENT PUBLICATION TRENDS FOR POULTRY DIAGNOSTICS
- 6.3.2 JURISDICTION & TOP APPLICANT ANALYSIS
- 6.4 FUTURE APPLICATIONS
- 6.4.1 PRECISION FLOCK-LEVEL DISEASE PREDICTION
- 6.4.2 MINI FULLY INTEGRATED ON-FARM MOLECULAR TESTING PLATFORMS
- 6.4.3 CONTINUOUS ENVIRONMENTAL & WASTEWATER MONITORING SYSTEMS
- 6.4.4 MULTIPLEX & SYNDROMIC POULTRY DIAGNOSTIC PANELS
- 6.4.5 SMART BIOSENSOR-ENABLED REAL-TIME HEALTH MONITORING
- 6.5 IMPACT OF AI/GENERATIVE AI ON POULTRY DIAGNOSTICS MARKET
- 6.5.1 INTRODUCTION
- 6.5.2 MARKET POTENTIAL IN POULTRY DIAGNOSTIC ECOSYSTEM
- 6.5.3 AI USE CASES
- 6.5.4 KEY COMPANIES IMPLEMENTING AI IN POULTRY DIAGNOSTICS MARKET
7 SUSTAINABILITY & REGULATORY LANDSCAPE
- 7.1 REGIONAL REGULATIONS & COMPLIANCE
- 7.1.1 REGULATORY ANALYSIS
- 7.1.2 REGULATORY BODIES, GOVERNMENT AGENCIES, AND OTHER ORGANIZATIONS
- 7.1.3 INDUSTRY STANDARDS
- 7.2 SUSTAINABILITY INITIATIVES
- 7.2.1 RECYCLED AND ECO-FRIENDLY MATERIALS FOR POULTRY DIAGNOSTIC PRODUCTS
- 7.2.2 SUSTAINABILITY IMPACT & REGULATORY POLICY INITIATIVES
- 7.2.3 CERTIFICATIONS, LABELING, AND ECO-STANDARDS
8 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.1 KEY STAKEHOLDERS & BUYING CRITERIA
- 8.1.1 KEY STAKEHOLDERS IN BUYING PROCESS
- 8.1.2 BUYING CRITERIA
- 8.2 CUSTOMER LANDSCAPE & BUYER BEHAVIOR
- 8.2.1 DECISION-MAKING PROCESS
- 8.2.2 ADOPTION BARRIERS & INTERNAL CHALLENGES
- 8.2.3 UNMET NEEDS FROM VARIOUS END-USE INDUSTRIES
- 8.2.4 MARKET PROFITABILITY
9 POULTRY DIAGNOSTICS MARKET, BY PRODUCT TYPE
- 9.1 INTRODUCTION
- 9.2 CONSUMABLES & REAGENTS
- 9.2.1 ELISA KITS & REAGENTS
- 9.2.1.1 Large-scale serological surveillance and vaccination monitoring to sustain demand
- 9.2.1.2 Global volume analysis of ELISA kits & reagents, 2024-2031 (thousand units)
- 9.2.2 PCR KITS & REAGENTS
- 9.2.2.1 Rising adoption of molecular diagnostics to accelerate demand
- 9.2.2.2 Global volume analysis of PCR kits & reagents, 2024-2031 (thousand units)
- 9.2.3 SAMPLE PREPARATION & EXTRACTION KITS
- 9.2.3.1 Increasing use of molecular testing to fuel market growth
- 9.2.3.2 Global volume analysis of sample preparation & extraction kits, 2024-2031 (thousand units)
- 9.2.4 CONTROLS & LABORATORY CONSUMABLES
- 9.2.4.1 Expanding diagnostic test volumes to drive consistent demand
- 9.2.4.2 Global volume analysis of controls & laboratory consumables, 2024-2031 (thousand units)
- 9.2.1 ELISA KITS & REAGENTS
- 9.3 INSTRUMENTS
- 9.3.1 PCR INSTRUMENTS
- 9.3.1.1 Growing need for rapid and high-sensitivity pathogen detection to boost market
- 9.3.1.2 Global volume analysis of PCR instruments, 2024-2031 (thousand units)
- 9.3.2 ELISA READERS & WASHERS
- 9.3.2.1 High-throughput serological testing to support continued use of ELISA readers
- 9.3.2.2 Global volume analysis of ELISA readers & washers, 2024-2031 (thousand units)
- 9.3.3 NUCLEIC ACID EXTRACTION SYSTEMS
- 9.3.3.1 Rising molecular testing workflows to drive adoption
- 9.3.3.2 Global volume analysis of nucleic acid extraction systems, 2024-2031 (thousand units)
- 9.3.4 OTHER LABORATORY AUTOMATION INSTRUMENTS
- 9.3.4.1 Global volume analysis of other laboratory automation instruments, 2024-2031 (thousand units)
- 9.3.1 PCR INSTRUMENTS
10 POULTRY DIAGNOSTICS MARKET, BY TEST TYPE
- 10.1 INTRODUCTION
- 10.2 PCR-BASED DIAGNOSTICS
- 10.2.1 AVIAN INFLUENZA PCR PANELS
- 10.2.1.1 Global surveillance programs and outbreak monitoring to boost demand
- 10.2.1.2 Global volume analysis of avian influenza PCR panels, 2024-2031 (thousand tests)
- 10.2.1.3 Global volume analysis of avian influenza PCR panels, 2024-2031 (thousand animals)
- 10.2.2 NEWCASTLE DISEASE PCR PANELS
- 10.2.2.1 High transmissibility of Newcastle disease to increase reliance on molecular PCR detection
- 10.2.2.2 Global volume analysis of Newcastle disease PCR panels, 2024-2031 (thousand tests)
- 10.2.2.3 Global volume analysis of Newcastle disease PCR panels, 2024-2031 (thousand animals)
- 10.2.3 ENDEMIC VIRAL PCR PANELS
- 10.2.3.1 Routine flock health monitoring to accelerate use of multiplex endemic viral PCR panels
- 10.2.3.2 Global volume analysis of endemic viral PCR panels, 2024-2031 (thousand tests)
- 10.2.3.3 Global volume analysis of endemic viral PCR panels, 2024-2031 (thousand animals)
- 10.2.4 MYCOPLASMA PCR PANELS
- 10.2.4.1 Need for early identification of mycoplasma infections to drive adoption
- 10.2.4.2 Global volume analysis of Mycoplasma PCR panels, 2024-2031 (thousand tests)
- 10.2.4.3 Global volume analysis of Mycoplasma PCR panels, 2024-2031 (thousand animals)
- 10.2.5 BACTERIAL PCR PANELS
- 10.2.5.1 Increasing focus on pathogen detection and food safety to support market growth
- 10.2.5.2 Global volume analysis of bacterial PCR panels, 2024-2031 (thousand tests)
- 10.2.5.3 Global volume analysis of bacterial PCR panels, 2024-2031 (thousand animals)
- 10.2.1 AVIAN INFLUENZA PCR PANELS
- 10.3 ELISA-BASED DIAGNOSTICS
- 10.3.1 ENDEMIC VIRAL ELISA TESTS
- 10.3.1.1 Routine immunity monitoring in poultry flocks to drive demand
- 10.3.1.2 Global volume analysis of endemic viral ELISA tests, 2024-2031 (thousand tests)
- 10.3.1.3 Global volume analysis of endemic viral ELISA tests, 2024-2031 (thousand animals)
- 10.3.2 AVIAN INFLUENZA ELISA TESTS
- 10.3.2.1 Large-scale serological surveillance programs to support use of avian influenza ELISA assays
- 10.3.2.2 Global volume analysis of avian influenza ELISA tests, 2024-2031 (thousand tests)
- 10.3.2.3 Global volume analysis of avian influenza ELISA tests, 2024-2031 (thousand animals)
- 10.3.3 NEWCASTLE DISEASE ELISA TESTS
- 10.3.3.1 Vaccination monitoring and flock immunity assessment to fuel demand
- 10.3.3.2 Global volume analysis of Newcastle disease ELISA tests, 2024-2031 (thousand tests)
- 10.3.3.3 Global volume analysis of Newcastle disease ELISA tests, 2024-2031 (thousand animals)
- 10.3.4 MYCOPLASMA ELISA TESTS
- 10.3.4.1 Ongoing flock health surveillance to increase adoption of mycoplasma ELISA testing
- 10.3.4.2 Global volume analysis of mycoplasma ELISA tests, 2024-2031 (thousand tests)
- 10.3.4.3 Global volume analysis of mycoplasma ELISA tests, 2024-2031 (thousand animals)
- 10.3.5 BACTERIAL ELISA TESTS
- 10.3.5.1 Growing emphasis on bacterial disease control to support use of bacterial ELISA diagnostics
- 10.3.5.2 Global volume analysis of bacterial ELISA tests, 2024-2031 (thousand tests)
- 10.3.5.3 Global volume analysis of bacterial ELISA tests, 2024-2031 (thousand animals)
- 10.3.1 ENDEMIC VIRAL ELISA TESTS
- 10.4 OTHER DIAGNOSTIC TESTS
11 POULTRY DIAGNOSTICS MARKET, BY DISEASE TYPE
- 11.1 INTRODUCTION
- 11.2 AVIAN INFLUENZA
- 11.2.1 GLOBAL SURVEILLANCE PROGRAMS AND FREQUENT HPAI OUTBREAKS TO DRIVE DEMAND
- 11.3 NEWCASTLE DISEASE
- 11.3.1 HIGH CONTAGIOUSNESS AND ONGOING OUTBREAK RISKS TO FUEL MARKET GROWTH
- 11.4 INFECTIOUS BRONCHITIS
- 11.4.1 EMERGENCE OF MULTIPLE IBV VARIANTS TO INCREASE DEMAND FOR DIAGNOSTICS
- 11.5 AVIAN MYCOPLASMOSIS
- 11.5.1 RISING FOCUS ON RESPIRATORY DISEASE CONTROL TO BOOST ADOPTION
- 11.6 SALMONELLOSIS
- 11.6.1 INCREASING FOOD SAFETY REGULATIONS AND SURVEILLANCE PROGRAMS TO BOOST MARKET GROWTH
- 11.7 INFECTIOUS BURSAL DISEASE
- 11.7.1 NEED TO MONITOR IMMUNOSUPPRESSIVE VIRAL INFECTIONS TO DRIVE DEMAND
- 11.8 OTHER INFECTIOUS DISEASES
12 POULTRY DIAGNOSTICS MARKET, BY POULTRY TYPE
- 12.1 INTRODUCTION
- 12.2 PCR-BASED DIAGNOSTICS
- 12.2.1 BROILERS
- 12.2.1.1 High-density meat production systems to drive strong adoption of PCR testing
- 12.2.2 LAYERS
- 12.2.2.1 Need for rapid detection of diseases affecting egg production to support market growth
- 12.2.3 BREEDERS
- 12.2.3.1 Prevention of vertical pathogen transmission to drive extensive PCR surveillance
- 12.2.4 TURKEYS
- 12.2.4.1 Disease monitoring in commercial turkey production to support adoption
- 12.2.5 DUCKS & GEESE
- 12.2.5.1 Role of waterfowl in avian influenza surveillance to increase PCR testing
- 12.2.6 OTHER POULTRY
- 12.2.1 BROILERS
- 12.3 ELISA-BASED DIAGNOSTICS
- 12.3.1 BROILERS
- 12.3.1.1 Monitoring vaccine-induced immunity to drive ELISA-based serological testing
- 12.3.2 LAYERS
- 12.3.2.1 Long production cycles and egg yield monitoring to support market growth
- 12.3.3 BREEDERS
- 12.3.3.1 Routine antibody monitoring for disease-free breeding programs to drive ELISA testing
- 12.3.4 TURKEYS
- 12.3.4.1 Need for effective vaccination monitoring to support ELISA-based diagnostics
- 12.3.5 DUCKS & GEESE
- 12.3.5.1 Serological surveillance of waterfowl populations to boost market growth
- 12.3.6 OTHER POULTRY
- 12.3.1 BROILERS
- 12.4 OTHER DIAGNOSTIC TESTS
- 12.4.1 BROILERS
- 12.4.1.1 Rapid screening and confirmatory testing to require conventional diagnostics
- 12.4.2 LAYERS
- 12.4.2.1 Long-term flock monitoring to drive continued use of traditional diagnostic tests
- 12.4.3 BREEDERS
- 12.4.3.1 Established serological methods to support disease surveillance in breeder flocks
- 12.4.4 TURKEYS
- 12.4.4.1 Routine disease screening and pathogen identification to sustain use of conventional diagnostics
- 12.4.5 DUCKS & GEESE
- 12.4.5.1 Surveillance of waterfowl-associated pathogens to maintain use of traditional diagnostic methods
- 12.4.6 OTHER POULTRY
- 12.4.1 BROILERS
13 POULTRY DIAGNOSTICS MARKET, BY SAMPLE TYPE
- 13.1 INTRODUCTION
- 13.2 SWABS
- 13.2.1 EASE OF COLLECTION AND SUITABILITY FOR MOLECULAR TESTING TO DRIVE WIDESPREAD USE OF SWAB SAMPLES
- 13.3 SERUM/BLOOD
- 13.3.1 GROWING FOCUS ON FLOCK IMMUNITY MONITORING AND VACCINATION ASSESSMENT TO SUPPORT DEMAND
- 13.4 TISSUE SAMPLES
- 13.4.1 NEED FOR CONFIRMATORY DIAGNOSIS AND DETAILED PATHOLOGICAL INVESTIGATION TO DRIVE MARKET GROWTH
- 13.5 ENVIRONMENTAL & FECAL SAMPLES
- 13.5.1 RISING EMPHASIS ON FARM-LEVEL SURVEILLANCE AND BIOSECURITY MONITORING TO BOOST ADOPTION
14 POULTRY DIAGNOSTICS MARKET, BY END USER
- 14.1 INTRODUCTION
- 14.2 VETERINARY DIAGNOSTIC LABORATORIES
- 14.2.1 CENTRAL ROLE IN DISEASE SURVEILLANCE AND HIGH TESTING VOLUMES TO DRIVE DEMAND
- 14.3 INTEGRATED POULTRY PRODUCERS
- 14.3.1 GROWING VERTICAL INTEGRATION AND NEED FOR CONTINUOUS FLOCK MONITORING TO INCREASE DIAGNOSTIC ADOPTION
- 14.4 GOVERNMENT & REFERENCE LABORATORIES
- 14.4.1 NATIONAL DISEASE SURVEILLANCE PROGRAMS AND REGULATORY TESTING REQUIREMENTS TO BOOST MARKET GROWTH
- 14.5 ACADEMIC & RESEARCH INSTITUTIONS
- 14.5.1 RISING RESEARCH ON POULTRY PATHOGENS AND DIAGNOSTIC INNOVATION TO SUPPORT DEMAND
- 14.6 OTHER SPECIALIZED LABORATORIES
- 14.6.1 INCREASING OUTSOURCING OF ADVANCED TESTING SERVICES TO DRIVE DEMAND
15 POULTRY DIAGNOSTICS MARKET, BY REGION
- 15.1 INTRODUCTION
- 15.2 NORTH AMERICA
- 15.2.1 MACROECONOMIC OUTLOOK FOR NORTH AMERICA
- 15.2.2 NORTH AMERICA: VOLUME ANALYSIS, BY PRODUCT TYPE, 2024-2031
- 15.2.3 NORTH AMERICA: VOLUME ANALYSIS, BY TEST TYPE, 2024-2031
- 15.2.4 US
- 15.2.4.1 Large-scale poultry production to drive demand for poultry diagnostic testing
- 15.2.5 CANADA
- 15.2.5.1 Strong biosecurity regulations and expanding poultry production to drive market
- 15.3 EUROPE
- 15.3.1 MACROECONOMIC OUTLOOK FOR EUROPE
- 15.3.2 EUROPE: VOLUME ANALYSIS, BY PRODUCT TYPE, 2024-2031
- 15.3.3 EUROPE: VOLUME ANALYSIS, BY TEST TYPE, 2024-2031
- 15.3.4 GERMANY
- 15.3.4.1 Germany to dominate European market during forecast period
- 15.3.5 UK
- 15.3.5.1 Robust disease surveillance programs to drive market growth
- 15.3.6 FRANCE
- 15.3.6.1 Frequent avian influenza outbreaks and large poultry production systems to fuels market growth
- 15.3.7 ITALY
- 15.3.7.1 Growing poultry production to support market growth
- 15.3.8 SPAIN
- 15.3.8.1 Expanding broiler production and increasing disease surveillance to drive market growth
- 15.3.9 NETHERLANDS
- 15.3.9.1 Advanced veterinary surveillance infrastructure to drive market growth
- 15.3.10 POLAND
- 15.3.10.1 Rapid expansion of poultry production and export-oriented farming to increase demand
- 15.3.11 RUSSIA
- 15.3.11.1 Expanding domestic poultry production to drive market growth
- 15.3.12 REST OF EUROPE
- 15.4 ASIA PACIFIC
- 15.4.1 MACROECONOMIC OUTLOOK FOR ASIA PACIFIC
- 15.4.2 ASIA PACIFIC: VOLUME ANALYSIS, BY PRODUCT TYPE, 2024-2031
- 15.4.3 ASIA PACIFIC: VOLUME ANALYSIS, BY TEST TYPE, 2024-2031
- 15.4.4 CHINA
- 15.4.4.1 China to dominate APAC market during forecast period
- 15.4.5 JAPAN
- 15.4.5.1 Recurring avian influenza outbreaks to drive market
- 15.4.6 INDIA
- 15.4.6.1 Rapidly expanding poultry industry and increasing disease surveillance initiatives to boost market growth
- 15.4.7 AUSTRALIA
- 15.4.7.1 Strong biosecurity regulations and advanced poultry production systems to support market growth
- 15.4.8 SOUTH KOREA
- 15.4.8.1 Frequent avian influenza outbreaks and intensive poultry farming to boost market growth
- 15.4.9 THAILAND
- 15.4.9.1 Export-oriented poultry production to drive market
- 15.4.10 NEW ZEALAND
- 15.4.10.1 Strict biosecurity systems and preventive disease surveillance programs to support market growth
- 15.4.11 INDONESIA
- 15.4.11.1 Large poultry population to fuel market growth
- 15.4.12 PHILIPPINES
- 15.4.12.1 Expanding poultry production and rising disease monitoring efforts to drive adoption
- 15.4.13 REST OF ASIA PACIFIC
- 15.5 LATIN AMERICA
- 15.5.1 MACROECONOMIC OUTLOOK FOR LATIN AMERICA
- 15.5.2 LATIN AMERICA: VOLUME ANALYSIS, BY PRODUCT TYPE, 2024-2031
- 15.5.3 LATIN AMERICA: VOLUME ANALYSIS, BY TEST TYPE, 2024-2031
- 15.5.4 BRAZIL
- 15.5.4.1 Large-scale poultry production and export-driven poultry industry to drive demand
- 15.5.5 MEXICO
- 15.5.5.1 Strong domestic poultry consumption to contribute to market growth
- 15.5.6 ARGENTINA
- 15.5.6.1 Expanding poultry production and increased disease monitoring to drive market
- 15.5.7 REST OF LATIN AMERICA
- 15.6 MIDDLE EAST & AFRICA
- 15.6.1 MACROECONOMIC OUTLOOK FOR MIDDLE EAST & AFRICA
- 15.6.2 MIDDLE EAST & AFRICA: VOLUME ANALYSIS, BY PRODUCT TYPE, 2024-2031
- 15.6.3 MIDDLE EAST & AFRICA: VOLUME ANALYSIS, BY TEST TYPE, 2024-2031
- 15.6.4 GCC COUNTRIES
- 15.6.4.1 Kingdom of Saudi Arabia (KSA)
- 15.6.4.1.1 Expanding domestic poultry production and national food security initiatives to boost growth
- 15.6.4.2 United Arab Emirates (UAE)
- 15.6.4.2.1 Increasing investments in poultry farming to fuel UAE's market growth
- 15.6.4.3 Rest of GCC Countries
- 15.6.4.1 Kingdom of Saudi Arabia (KSA)
- 15.6.5 REST OF MIDDLE EAST & AFRICA
16 COMPETITIVE LANDSCAPE
- 16.1 OVERVIEW
- 16.2 KEY PLAYER STRATEGY/RIGHT TO WIN
- 16.3 REVENUE ANALYSIS, 2021-2025
- 16.4 MARKET SHARE ANALYSIS, 2025
- 16.4.1 US MARKET SHARE ANALYSIS, 2025
- 16.4.2 EUROPE MARKET SHARE ANALYSIS, 2025
- 16.4.3 RANKING OF KEY MARKET PLAYERS
- 16.5 COMPANY EVALUATION MATRIX: KEY PLAYERS, 2025
- 16.5.1 STARS
- 16.5.2 EMERGING LEADERS
- 16.5.3 PERVASIVE PLAYERS
- 16.5.4 PARTICIPANTS
- 16.5.5 COMPANY FOOTPRINT: KEY PLAYERS, 2025
- 16.5.5.1 Company footprint
- 16.5.5.2 Region footprint
- 16.5.5.3 Product type footprint
- 16.5.5.4 Test type footprint
- 16.5.5.5 Disease type footprint
- 16.6 COMPANY EVALUATION MATRIX: STARTUPS/SMES, 2025
- 16.6.1 PROGRESSIVE COMPANIES
- 16.6.2 RESPONSIVE COMPANIES
- 16.6.3 DYNAMIC COMPANIES
- 16.6.4 STARTING BLOCKS
- 16.6.5 COMPETITIVE BENCHMARKING: STARTUPS/SMES, 2025
- 16.6.5.1 Detailed list of key startups/SMEs
- 16.6.5.2 Competitive benchmarking of key startups
- 16.7 BRAND/PRODUCT COMPARISON
- 16.8 R&D EXPENDITURE OF KEY PLAYERS
- 16.9 COMPANY VALUATION & FINANCIAL METRICS
- 16.9.1 FINANCIAL METRICS
- 16.9.2 COMPANY VALUATION
- 16.10 COMPETITIVE SCENARIO
- 16.10.1 PRODUCT LAUNCHES
- 16.10.2 DEALS
- 16.10.3 EXPANSIONS
- 16.10.4 OTHER DEVELOPMENTS
17 COMPANY PROFILES
- 17.1 KEY PLAYERS
- 17.1.1 IDEXX LABORATORIES, INC.
- 17.1.1.1 Business overview
- 17.1.1.2 Products offered
- 17.1.1.3 MnM view
- 17.1.1.3.1 Key strengths
- 17.1.1.3.2 Strategic choices
- 17.1.1.3.3 Weaknesses & competitive threats
- 17.1.2 THERMO FISHER SCIENTIFIC INC.
- 17.1.2.1 Business overview
- 17.1.2.2 Products offered
- 17.1.2.3 MnM view
- 17.1.2.3.1 Key strengths
- 17.1.2.3.2 Strategic choices
- 17.1.2.3.3 Weaknesses & competitive threats
- 17.1.3 ZOETIS SERVICES LLC
- 17.1.3.1 Business overview
- 17.1.3.2 Products offered
- 17.1.3.3 Recent developments
- 17.1.3.3.1 Deals
- 17.1.3.3.2 Expansions
- 17.1.3.3.3 Other developments
- 17.1.3.4 MnM view
- 17.1.3.4.1 Key strengths
- 17.1.3.4.2 Strategic choices
- 17.1.3.4.3 Weaknesses & competitive threats
- 17.1.4 BIOCHEK
- 17.1.4.1 Business overview
- 17.1.4.2 Products offered
- 17.1.4.3 Recent developments
- 17.1.4.3.1 Product launches
- 17.1.4.3.2 Deals
- 17.1.4.4 MnM view
- 17.1.4.4.1 Key strengths
- 17.1.4.4.2 Strategic choices
- 17.1.4.4.3 Weaknesses & competitive threats
- 17.1.5 INNOVATIVE DIAGNOSTICS
- 17.1.5.1 Business overview
- 17.1.5.2 Products offered
- 17.1.5.3 Recent developments
- 17.1.5.3.1 Other developments
- 17.1.5.4 MnM view
- 17.1.5.4.1 Key strengths
- 17.1.5.4.2 Strategic choices
- 17.1.5.4.3 Weaknesses & competitive threats
- 17.1.6 BIONOTE
- 17.1.6.1 Business overview
- 17.1.6.2 Products offered
- 17.1.6.3 Recent developments
- 17.1.6.3.1 Deals
- 17.1.7 BIO-RAD LABORATORIES, INC.
- 17.1.7.1 Business overview
- 17.1.7.2 Products offered
- 17.1.8 GD
- 17.1.8.1 Business overview
- 17.1.8.2 Products offered
- 17.1.8.3 Recent developments
- 17.1.8.3.1 Product launches
- 17.1.8.3.2 Deals
- 17.1.8.3.3 Expansions
- 17.1.8.3.4 Other developments
- 17.1.9 INDICAL BIOSCIENCE GMBH
- 17.1.9.1 Business overview
- 17.1.9.2 Products offered
- 17.1.9.3 Recent developments
- 17.1.9.3.1 Product enhancements
- 17.1.10 SHENZHEN BIOEASY BIOTECHNOLOGY, INC.
- 17.1.10.1 Business overview
- 17.1.10.2 Products offered
- 17.1.11 RING BIOTECHNOLOGY CO., LTD.
- 17.1.11.1 Business overview
- 17.1.11.2 Products offered
- 17.1.11.3 Recent developments
- 17.1.11.3.1 Product launches
- 17.1.12 BIO-X DIAGNOSTICS S.A.
- 17.1.12.1 Business overview
- 17.1.12.2 Products offered
- 17.1.13 MEGACOR DIAGNOSTIK GMBH
- 17.1.13.1 Business overview
- 17.1.13.2 Products offered
- 17.1.14 GOLD STANDARD DIAGNOSTICS
- 17.1.14.1 Business overview
- 17.1.14.2 Products offered
- 17.1.14.2.1 Other developments
- 17.1.15 BIONEER CORPORATION
- 17.1.15.1 Business overview
- 17.1.15.2 Products offered
- 17.1.1 IDEXX LABORATORIES, INC.
- 17.2 OTHER PLAYERS
- 17.2.1 ELABSCIENCE, INC.
- 17.2.2 BIOSELLAL
- 17.2.3 BIOGAL
- 17.2.4 BIOSTONE ANIMAL HEALTH
- 17.2.5 UBIO BIOTECHNOLOGY SYSTEMS PVT. LTD.
- 17.2.6 ABBEXA
- 17.2.7 BIOINGENTECH
- 17.2.8 AFFIGEN INC.
- 17.2.9 ALVEO TECHNOLOGIES
- 17.2.10 HIMEDIA LABORATORIES
18 RESEARCH METHODOLOGY
- 18.1 RESEARCH DATA
- 18.2 RESEARCH METHODOLOGY DESIGN
- 18.2.1 SECONDARY DATA
- 18.2.1.1 Key data from secondary sources
- 18.2.2 PRIMARY DATA
- 18.2.2.1 Key data from primary sources
- 18.2.2.2 Key industry insights
- 18.2.1 SECONDARY DATA
- 18.3 MARKET SIZE ESTIMATION
- 18.3.1 BOTTOM-UP APPROACH
- 18.4 MARKET BREAKDOWN & DATA TRIANGULATION
- 18.5 MARKET SHARE ANALYSIS
- 18.5.1 RESEARCH ASSUMPTIONS
- 18.5.2 GROWTH RATE ASSUMPTIONS
- 18.6 RISK ASSESSMENT
- 18.7 RESEARCH LIMITATIONS
- 18.7.1 METHODOLOGY-RELATED LIMITATIONS
- 18.7.2 SCOPE-RELATED LIMITATIONS
19 APPENDIX
- 19.1 DISCUSSION GUIDE
- 19.2 KNOWLEDGESTORE: MARKETSANDMARKETS' SUBSCRIPTION PORTAL
- 19.3 CUSTOMIZATION OPTIONS
- 19.3.1 PRODUCT ANALYSIS
- 19.3.2 COMPANY INFORMATION
- 19.3.3 GEOGRAPHIC ANALYSIS
- 19.3.4 REGIONAL/COUNTRY-LEVEL MARKET SHARE ANALYSIS
- 19.3.5 COUNTRY-LEVEL VOLUME ANALYSIS, BY PRODUCT TYPE
- 19.3.6 MARKET SHARE ANALYSIS, BY PRODUCT TYPE (TOP 5 PLAYERS)
- 19.3.7 ANY CONSULT/CUSTOM REQUIREMENTS AS PER CLIENT REQUESTS
- 19.4 RELATED REPORTS
- 19.5 AUTHOR DETAILS





